The development and formulation of quantum physics coincided with the consolidation of theoretical physics as a subdiscipline, the existence of which was the result of a long process whose origins go back at least to the middle of the nineteenth century. Throughout this period, it acquired characteristics, which, even if not original, distinguished it from earlier ways of theorizing about the physical world. Especially as it developed in the German-speaking world, this new theoretical physics emphasized mathematical laws and more significantly a mathematical deduction of quantitative rules from a few assumptions and the connection of these rules to empirical findings. While valuing the empirical examination of these rules, the new theoretical physics reinforced, and sometimes even created, a division of labor within physics between experimentalists and theoreticians. In this aspect, as in many others, this kind of theoretical physics became dominant in the twentieth century across linguistic and national borders.
Historians have pointed to a connection between the development of quantum mechanics on its elaborated mathematical edifice and the rise of the new theoretical physics. For example, in their classic history of the subdiscipline in Germany, Christa Jungnickel and Russell McCormmach write:
Theoretical physics experienced some of its greatest advances, and German theoretical physicists played a significant and often leading part in [the special and general theories of relativity and the early quantum theory]. (Jungnickel and McCormmach 1986, 304)
That theoretical physics played a leading role in the development of quantum physics, however, meant neither that the more traditional “experimental physics” stopped contributing to the process, nor that its participants confined their role to the testing of theories. On the contrary, as I illustrate below, they also added to the theoretical understanding of the microphysical world. The contribution of this kind of experimental physics seems to be especially significant with the developments that took place before the formulation of matrix and wave mechanics and particularly in new research areas, like photoelectricity and X-rays.
Physicists working in the more traditional style of research designed and performed experiments to explore and later understand various aspects of newly observed effects. For example, in photoelectricity many experimentalists strove to determine the source and nature of the charge carriers, leading eventually to the identification of the latter with electrons. Experimentalists took on the task of clarifying the phenomena related to the new effects and entities and to determine their characteristics (e.g., the kind of irradiated metal, the intensity, direction, polarization and wavelength of the light, and temperature). They suggested a number of hypotheses and explanations to account for the new phenomena. Yet contrary to the new theoretical physics, the kind of theoretical thought associated with experimental physics rarely involved either elaborated mathematical derivations from basic laws or exact quantitative rules for empirical test. There was no systematic theory to describe and organize the observed phenomena. Thus, experiment and theory were closely intertwined, often within the work of the same person, creating situations in which empirical investigation aimed at examining a particular hypothesis, and assumptions followed particular experimental results. Experiments were frequently explorative in character (Steinle 1998), and were used directly to supply new information and to decide between competing claims and hypotheses. Even when more encompassing hypotheses were suggested, experiments directly examined their implications, rather than a mathematical rule deduced from them.
Unlike this situation, in the ideal type of theoretical physics, experiments are confined to test theoretically derived quantitative rules. Black-body radiation provides a good example. Such experts in exact measurement as Heinrich Rubens, Ferdinand Kurlbaum, Otto Lummer and Ernst Pringsheim determined the exact mathematical curve of the wavelength distribution of black-body radiation. Their precise measurements tested the exact form of the mathematical relations but were detached from the physical assumptions made in deriving this quantitative rule. Atomic spectra following Niels Bohr’s atom provides another example of measurements as a way to test an elaborate theory as a whole. The atomic and molecular emission lines became meaningful for physical claims only by comparing them with the quantitative results of the theories of Bohr and Arnold Sommerfeld. In its ideal type, an empirical deviation from a theoretical law calls for a change in the assumptions of the theory, a change that could lead to a new rule fitting the experimental results. The new rule should consequently be compared with further, more precise or extended measurements, or to measurements of other related phenomena. Sommerfeld’s modification of, and addition to, Bohr’s atomic theory to improve its match with measurements showing fine structure of spectral lines is an example of this kind of study.
Thus, theoretical physics of this kind supposes a clear division between theorization and experimentation. This separation is logical, chronological and often also personal: they are different ways of doing science, they take place at different times and are often performed by different actors. However, in the early twentieth century, both traditional experimental physics and most of the physical research outside the German cultural sphere did not assume such a clear separation. Experiments contributed to theorizing by providing information about various aspects of phenomena, as well as examining the implications of specific hypotheses, and not only of one particular quantitative rule. Moreover, experimentalists suggested many hypotheses, concepts and theories while designing their experiments and reflecting on them. In Germany, the practitioners of this kind of laboratory research were called “experimental physicists,” while their colleagues who worked at precise tests of quantitative rules were coined “measuring physicists.” Measuring physics was the laboratory endeavor associated with theoretical physics (Jungnickel and McCormmach 1986, 120). In the late nineteenth century, exact measurements were the only laboratory research still practiced by German theoretical physicists. With the younger generation of theoreticians developing quantum physics, however, the separation between physicists deriving quantitive expressions and those who empirically tested them became almost complete. In other cultural realms, including the English- and the French-speaking worlds, the division between theoreticians and experimentalists emerged later. So, in the early decades of the twentieth century in these countries, physicists engaged with theory often conducted explorative and more traditional kinds of experiments. In this paper, following the German terminology of the time, this kind of research I call experimental physics.
This article illustrates how physicists working in this more traditional kind of experimental physics developed and examined new theoretical understandings of phenomena relating to ultraviolet radiation, Röntgen (X) rays and
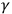 -rays, and their interactions with matter. Although unexpected, the discoveries of the photoelectric effect (1886) and X-rays (1895) did not seem to threaten the foundations of contemporary physics. Only further exploration of these phenomena at the beginning of the twentieth century by “experimental physicists” revealed contradictions with the common (later called “classical”) laws and concepts of physics. Conclusions, drawn from many experiments in this tradition, and not solely independent results, convinced many scientists of the need to adopt assumptions that contradicted classical physics. Moreover, new notions that emerged in this kind of research evolved into later concepts of quantum physics. In retrospects, “quantum hypotheses” that assume discrete distribution of energy were the most important among these.
-rays, and their interactions with matter. Although unexpected, the discoveries of the photoelectric effect (1886) and X-rays (1895) did not seem to threaten the foundations of contemporary physics. Only further exploration of these phenomena at the beginning of the twentieth century by “experimental physicists” revealed contradictions with the common (later called “classical”) laws and concepts of physics. Conclusions, drawn from many experiments in this tradition, and not solely independent results, convinced many scientists of the need to adopt assumptions that contradicted classical physics. Moreover, new notions that emerged in this kind of research evolved into later concepts of quantum physics. In retrospects, “quantum hypotheses” that assume discrete distribution of energy were the most important among these.
Theoretical arguments did play a crucial role in other important strands that led to quantum theory. Most famously, Max Planck needed to introduce the quantum hypothesis as a theoretical procedure in statistical thermodynamics (regardless of his own earlier “classical” interpretation of it) (Darrigol 1992). While exact measurements showed the need to change the law of black-body radiation, only a theoretical derivation of the law revealed the need to introduce the quantum hypothesis. Moreover, in 1905, Albert Einstein produced his more radical hypothesis of light quanta based on an argument that revealed a conflict between the theories of electrodynamics and thermodynamics, without evoking any novel empirical findings (Büttner et.al. 2003). This kind of theoretical scrutiny, however, could not lead to similar results regarding the interaction of radiation with matter. Rather, experimental research discerned limitations of and contradictions with a conventional (classical) explanation of ultraviolet radiation, and X- and
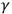 -rays, and their interaction with matter. Around 1911–1913, the combination of evidence from these phenomena convinced most physicists to admit discontinuity in radiation phenomena, usually in the form of a quantum hypothesis, which they connected to theoretical findings about black-body radiation.
-rays, and their interaction with matter. Around 1911–1913, the combination of evidence from these phenomena convinced most physicists to admit discontinuity in radiation phenomena, usually in the form of a quantum hypothesis, which they connected to theoretical findings about black-body radiation.
1.1 The Nature of X- and
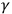 -Rays
-Rays
Wilhelm Röntgen’s discovery of X-rays triggered an extraordinary wave of experiments about their main properties and raised almost as many speculations about their nature. Most of these experiments studied the interaction of these rays with matter and electromagnetic radiation in the tradition of experimental physics, experiments that were mostly quantitative. Usually these experiments did not require the high precision needed in black-body measurements. Rough estimations and even an order of magnitude were often sufficient, at least in the early stages. Röntgen himself was a representative of experimental physics, and his famous discovery, like most of his research, belonged to that tradition (Katzir 2006b, 44). Within a few months, the experiments carried out in that tradition led to the general view that X-rays were some kind of electromagnetic waves. Experimentalists showed that the rays could penetrate matter (which was not characteristic of any particle), were not deflected by strong electric and magnetic fields (thus, the rays were not charged), and produced photographs on chemical paper (as did infrared, visible and ultraviolet light). Röntgen himself supposed that the rays were longitudinal or condensational ether waves, similar to acoustical waves in air, a kind of wave long sought for in continental electromagnetism, often suggested in the experimental research on cathode rays, and thus a natural assumption for Röntgen (Darrigol 2000, 284–287). James Maxwell’s theory, however, did not allow for longitudinal waves, therefore many physicists leaned toward the view that X-rays were ordinary light, i.e., transverse waves, of very high frequency. The electromagnetic assumptions led to attempts to polarize (as only transverse waves do) and diffract (as all waves do) the rays. Yet, early failures in producing polarization and diffraction did not refute the wave assumption, since such an assumption did not provide a prediction about the magnitude of the effects. In this early phase, the non-effect constrained the assumption, i.e. the wavelength of the X-rays, but did not lead to its rejection. The rays, however, also failed to show other features of regular waves like reflection and refraction. The impulse hypothesis, according to which X-rays were transverse electromagnetic waves of very short duration, offered a way out of the failure of X-rays to present these expected features.1
Cambridge mathematical physicist George Gabriel Stokes proposed the impulse hypothesis in 1896 as a theoretical idea within the classical electromagnetic wave theory. Still, its justification originated in qualitative findings about X-rays. It allowed for the combination of spatial continuity, characteristic of waves and fitting such an observation as neutral electricity and strong penetrating power, with the temporal discontinuity usually associated with particles, as suggested by the rays’ strength and short duration. Stokes and Joseph J. Thomson suggested a mathematical theory of the impulses and their interactions. Later in 1899, German theoretician Sommerfeld showed that the hypothesis was flexible enough to answer another direct challenge by experiment, namely X-ray diffraction (by broadening the image, without showing the interference pattern), which had been observed a few months earlier by Cornelis Wind and Hermanus Haga in Groningen. “In one form or another, the impulse hypothesis of X-rays achieved supremacy in the period 1903–1905.” (Wheaton 1983, 15–67, quotation on p. 48). Experiments on
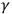 -rays, recently differentiated from
-rays, recently differentiated from
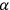 and
and
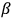 radioactive rays, pointed to a close similarity between them and X-rays. Consequently, most physicists regarded
radioactive rays, pointed to a close similarity between them and X-rays. Consequently, most physicists regarded
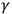 -rays as a stronger kind of electromagnetic impulse.
-rays as a stronger kind of electromagnetic impulse.
However, because it was a kind of wave assumption, the impulse hypothesis led to two major problems, which historian Bruce Wheaton in his important book on the experimental origins of particle-wave duality termed the paradoxes of quantity and quality. Since the impulse, as a kind of wave, propagates spherically, it should have ionized all atoms on its way. Yet, already in 1896 and 1897, Thomson and independently Ernst Dorn had found that only a tiny fraction of the atoms were ionized. This is the paradox of quantity. On the other hand, further experiments, especially on “secondary electrons” emitted by the absorption of X-rays, showed that X-rays communicated energy to individual electrons—of a similar magnitude to what was needed for their own generation, without any apparent weakening with distance.2 How a spherical propagation, like an impulse, could deliver its whole energy to a localized place in space was the paradox of quality (Wheaton 1983, 71–79). Notice that these experiments were not carried out to examine a particular mathematical prediction but to explore the behavior and nature of these rays, mainly independently of the impulse hypothesis. Although quantitative, these experiments did not require high precision (i.e., an order of magnitude was more than enough for the quantity paradox). This kind of semi qualitative results characterized experimental rather than measuring physics.
Faced with the paradoxes of quantity and quality, William Henry Bragg suggested in 1907 that X- and
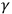 -rays were neutral pairs of an electron and a positive charge, rather than an impulse. Working alone in Australia, Bragg was a dissident. European physicists continued to think of the rays as wave impulses, or even as simple waves. Bragg’s suggestion was far from a formal theory but led him and Charles Barkla in Liverpool to do further experimental work. Barkla, however, wanted to challenge Bragg’s conclusions. Bragg and Barkla designed their experiments to check direct qualitative consequences of the wave and the corpuscular hypotheses, without the mediation of an elaborate theory. Barkla supported the wave point of view by obtaining partial polarization of X-rays (a property of transverse waves but not of particles), as well as showing the homogeneity of secondary X-rays. In his experiments on strong X-rays (i.e. X-rays of high penetrating power) and
-rays were neutral pairs of an electron and a positive charge, rather than an impulse. Working alone in Australia, Bragg was a dissident. European physicists continued to think of the rays as wave impulses, or even as simple waves. Bragg’s suggestion was far from a formal theory but led him and Charles Barkla in Liverpool to do further experimental work. Barkla, however, wanted to challenge Bragg’s conclusions. Bragg and Barkla designed their experiments to check direct qualitative consequences of the wave and the corpuscular hypotheses, without the mediation of an elaborate theory. Barkla supported the wave point of view by obtaining partial polarization of X-rays (a property of transverse waves but not of particles), as well as showing the homogeneity of secondary X-rays. In his experiments on strong X-rays (i.e. X-rays of high penetrating power) and
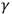 -rays, Bragg, on the other hand, demonstrated their particle-like properties. For example, he showed that the direction of the emitted secondary electrons depended on the direction of the
-rays, Bragg, on the other hand, demonstrated their particle-like properties. For example, he showed that the direction of the emitted secondary electrons depended on the direction of the
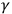 -rays that induced them. Their experiments and the controversy between the two physicists showed the difficulties of explaining the behavior of these rays either as particles or as waves (Wheaton 1983, 81–103; Stuewer 1971b).
-rays that induced them. Their experiments and the controversy between the two physicists showed the difficulties of explaining the behavior of these rays either as particles or as waves (Wheaton 1983, 81–103; Stuewer 1971b).
1.2 Photoelectricity and the Triggering Hypothesis
Photoelectricity originated in Heinrich Hertz’s experimental research program that led him to the production and detection of electromagnetic waves. In the winter of 1886–1887, he discovered that ultraviolet light enhances electric spark-discharge. In 1888, following Hertz, Wilhelm Hallwachs observed electric discharge from metal plates exposed to ultraviolet light in a vacuum tube. This effect became the paradigmatic example for photoelectricity. Experimentalists throughout Europe explored several aspects of this new phenomenon, examining the metals, charge carriers, gases, tubes and light that interact in these effects. Still, for more than twenty years it failed to attract much attention from theoretical physicists but remained the domain of experimental physics (Stuewer 1971a; Buchwald 1994, 243–244; Hallwachs 1916; von Schweidler 1898, 883–894).
In 1902, Phillip Lenard examined the relationship between the intensity of the incident light and the energy of the electric discharge. Exploiting recent experiments of his own and those carried out by Egon von Schweidler and Thomson that identified the carriers of the photoelectric current with electrons, Lenard measured the energy of the emitted electrons. Surprisingly, he found that their energy was independent from the intensity of light, i.e., from the power of the electromagnetic wave. He assumed, therefore, that the electrons did not leave the metal with energy received from the wave, but with energy they already possessed when they were inside the atom. Relying on Hendrik Antoon Lorentz’s picture of the electron as a virtual oscillator within the atom, Lenard assumed that the electromagnetic wave only triggered the release of electrons through some undetailed resonance between their and the electrons’ frequency of oscillation. Since the wave was not thought to provide energy to the electrons, this mechanism was termed the “triggering hypothesis” (von Schweidler 1898; Wheaton 1978; Heilbron 1994).
Lenard’s idea was very similar to the common explanation of anomalous dispersion as a resonance between light and matter, proposed by Wolfgang Sellmeier in 1872 (Sellmeier 1872; Jordi Taltavull forthcoming). It became the most popular answer to the paradox of quality. Since, according to the hypothesis, the phenomenon is atomistic in character, the triggering hypothesis also explained gas ionization by ultraviolet light. To support its extension to other phenomena, two of Thomson’s students examined empirical consequences of the hypothesis for
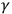 - and X-rays. In 1905, John A. McClelland found a correlation between atomic weight and the intensity of secondary electrons induced by
- and X-rays. In 1905, John A. McClelland found a correlation between atomic weight and the intensity of secondary electrons induced by
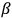 - and
- and
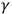 -rays. From his point of view, this correlation indicated that the electron’s energy originated in the atoms. Two years later, P. D. Innes showed that, as in the case of light, “[t]he velocity of the fastest electrons emitted from each metal [by X-rays] is completely independent of the intensity of the primary rays, but increases with the hardness of the tube [‘the penetrating power’ of the X-rays]” (Innes 1907, 462; Wheaton 1983, 73–75). Similar experiments in Germany and the United States led to the same conclusion. These results convinced many experimentalists that the energy of the electrons originated in the atoms, and thus that the triggering hypothesis could be extended to X- and
-rays. From his point of view, this correlation indicated that the electron’s energy originated in the atoms. Two years later, P. D. Innes showed that, as in the case of light, “[t]he velocity of the fastest electrons emitted from each metal [by X-rays] is completely independent of the intensity of the primary rays, but increases with the hardness of the tube [‘the penetrating power’ of the X-rays]” (Innes 1907, 462; Wheaton 1983, 73–75). Similar experiments in Germany and the United States led to the same conclusion. These results convinced many experimentalists that the energy of the electrons originated in the atoms, and thus that the triggering hypothesis could be extended to X- and
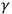 -rays.
The direct experimental corroboration of theoretical assumptions displays the close connection between theoretical thinking and the laboratory. Following its extension to X-rays, physicists also saw an answer to the paradox of quantity in the triggering hypothesis, assuming that at any instance only a small portion of the atoms contained electrons oscillating at the frequency of the ray (Innes 1907). By then, the triggering hypothesis was popular among German and British physicists alike.
-rays.
The direct experimental corroboration of theoretical assumptions displays the close connection between theoretical thinking and the laboratory. Following its extension to X-rays, physicists also saw an answer to the paradox of quantity in the triggering hypothesis, assuming that at any instance only a small portion of the atoms contained electrons oscillating at the frequency of the ray (Innes 1907). By then, the triggering hypothesis was popular among German and British physicists alike.
From the perspective of the new theoretical physics, measuring physicists should have tested the mathematical laws that followed from the triggering hypothesis, namely the relation between the frequency of light and the maximal electron velocity. This appears to be a classical case in which exact measurements should arbitrate between two alternative mathematical laws and consequently between the theories that produce them. According to Lenard’s assumption, the frequency of light is proportional to the velocity of the electrons, while, according to Einstein’s light quantum, the frequency is proportional to the square of the velocity. Unlike Lenard, who advanced a triggering mechanism as a generalization from and explanation of a particular experimental result, Einstein proposed the light quantum hypothesis in 1905 to answer a theoretical puzzle within statistical thermodynamics. Although he had not conceived the assumption of discrete quanta of light energy as the answer to any particular empirical findings, Einstein did employ it to explain the phenomena of fluorescence, ionization by ultraviolet light, and, more famously, photoelectricity (Einstein 1905; Klein 1963). Still, despite this apparently clear way to arbitrate between theories, experimentalists did not rush to test the predictions of the triggering hypothesis versus Einstein’s law for photoelectricity. When they did examine the triggering hypothesis, they first concentrated on other implications of the hypothesis, especially those that did not require high precision to distinguish between the two mathematical laws. They found other evidence more compelling. In other words, they preferred the methods of experimental physics to those of measuring physics associated with the new kind of theoretical physics.
In 1907, Erich Ladenburg carried out a series of experiments that examined the triggering hypothesis and its implications for the electrons inside matter. If this hypothesis was true, one could learn about the energy of electrons inside the atom (or in the metal) from the emission energy of photoelectrons. Ladenburg thought that, due to thermal motion, more electrons inside the matter should have high velocities with increasing temperature, and thus he examined the changes in photoelectricity at high temperatures (up to 800ºC). He found, however, no change in photoelectric current and suggested that the effect on the distribution of electron velocities was too small to be observed in the experiment. The implication of the result for the triggering hypothesis was far from decisive. Two years earlier, A. Lienhop, a student of Lenard, viewed the independence of the effect from temperature (which he showed for low temperature) as a corroboration of the hypothesis, since he assumed that the oscillations of electrons inside the atoms are independent of the temperature (Lienhop 1906).
For Ladenburg, however, the result indicated the need to examine the triggering hypothesis further. Thus, during 1907–1908, he examined the relationship between the frequency of absorbing light and the velocity of emitted electrons. He found that “the initial velocity of the electrons is proportional to the frequency of the absorbed light” (Ladenburg 1907, 514). While this conclusion agreed with the predication of the triggering hypothesis, his finding that light releases electrons in all the many frequencies examined cast doubts on it. To keep the triggering explanation, the latter finding required the assumption of many modes of electronic oscillations in matter. Ladenburg did not worry about that, probably because he considered that electronic oscillations in the metal happened also between the atoms and not only inside the atom. Others like Thomson, however, found it problematic to explain these results, especially as Thomson assumed that metal atoms consist of dozens rather than thousands of electrons (Wheaton 1978, 136–139). By 1910, further experiments corroborated Thomson’s view about the reduced number of electrons in the atom (Heilbron 1977), and therefore made the triggering hypothesis dubious.
Furthermore, soon after the publication of Ladenburg’s results, Abraham Joffé (1907) challenged their consistency with the triggering hypothesis. Against the assertion of the experimenter, he claimed that Ladenburg’s measurements did not establish a linear relation between maximum velocity and light frequency. Joffé showed that the results agreed at least as well with Einstein’s “atomistic hypothesis of the radiation energy” (Katzir 2006a, 452–453). In the narrow spectrum of ultraviolet light that could be employed to release electrons from metals, the difference between the two mathematical relations (i.e., between a straight line and a segment of a parabola) was smaller than the experimental error. Moreover, these were delicate experiments prone to quite a few sources of error. Further attempts to measure the relations between frequency and velocity led to conflicting and ambiguous results. The experiments could not arbitrate between the quantitative relations that followed the triggering and Einstein’s hypotheses. While a few physicists trusted two 1912 measurements, one by Owen Richardson and Karl Compton and another by Arthur Hughes, which supported Einstein’s relation, other experimentalists cast doubts on some of their results, and thereby, on their conclusions. The indecisive outcome of these experiments exemplifies the limitations of the measuring approach. Only Robert Millikan’s 1916 measurements settled the issue to the satisfaction of most experimentalists. Yet, most physicists had already abandoned the triggering hypothesis at the beginning of the decade. They had done so because of the accumulation of other evidence from different areas (Katzir 2006a, 453–454).
Lenard himself provided the strongest individual blow to the triggering hypothesis. In 1911, together with Carl Ramsauer, he looked for a correlation between the emission of electrons and the absorption of ultraviolet light, and thus of energy, in the ionization of gases. Hence, they designed the experiment to check the fundamental assumption of the triggering mechanism, i.e., that the energy of the electrons originated in matter. Against the triggering hypothesis, the experiment displayed a correlation between the absorption of light energy and electron emission, showing, in Ramsauer’s words, that “the energy of the ejected electron does not come from the atoms as originally assumed by Mr. Lenard, but from the absorbed light” (Wheaton 1983, 178). In the tradition of experimental physics, the experiment directly examined a theoretical assumption—the atomic source of the photoelectrons’ energy. This contradiction of the triggering hypothesis highlighted the paradoxes of radiations, paradoxes which became even more acute with the link made between ultraviolet light and X- and
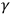 -rays.
-rays.
1.3 Visible Light and X-rays
One way to avoid, or at least to lessen, the contradictory aspects of radiation was to separate X- and
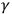 -rays from visible light and from ultraviolet light. In a sense, this was Bragg’s and Thomson’s strategy. Concentrating on the powerful radiation, Bragg preferred viewing X- and
-rays from visible light and from ultraviolet light. In a sense, this was Bragg’s and Thomson’s strategy. Concentrating on the powerful radiation, Bragg preferred viewing X- and
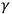 -rays as a neutral pair. After toying with the triggering hypothesis, Thomson had, by 1907, abandoned it. Instead, he looked for an answer in the structure of the electromagnetic ether. He viewed the ether as full of “Faraday tubes,” which consisted of bundles of lines of electric force giving a coarse-grained appearance to the ether, rather than a continuous one, when the plane of an electromagnetic wave intersected these tubes. These apparently grained surfaces would guide the X-rays impulses only in a discrete number of specific directions. Like Röntgen’s suggestion of longitudinal waves, Thomson developed the concept of tubes of force in his earlier experimental study of cathode rays and electric discharge (Smith 2001, 27–35; Navarro 2012, 60–70, 114–119). Yet, interference precluded Thomson from assuming that the apparent grainy character of the ether also influenced light.3 Instead, in 1910 and 1913 he advanced two incompatible but equally speculative atomic models that could explain the continuity in the velocity of the emitted electrons. Disregarding Lenard and Ramsauer’s result, both models involved a kind of triggering hypothesis. Suggesting tubes of force inside the atom, Thomson succeeded in regaining a threshold frequency for releasing an electron equivalent to that of Einstein’s equation. Despite the deep differences in their fundamental assumptions, some physicists conflated Thomson’s view of corpuscle-like radiation with Einstein’s, since both ascribed particle-like properties to radiation (Wheaton 1983, 136–142; Stuewer 1970, 252–253; Millikan 1963, 221–223).
-rays as a neutral pair. After toying with the triggering hypothesis, Thomson had, by 1907, abandoned it. Instead, he looked for an answer in the structure of the electromagnetic ether. He viewed the ether as full of “Faraday tubes,” which consisted of bundles of lines of electric force giving a coarse-grained appearance to the ether, rather than a continuous one, when the plane of an electromagnetic wave intersected these tubes. These apparently grained surfaces would guide the X-rays impulses only in a discrete number of specific directions. Like Röntgen’s suggestion of longitudinal waves, Thomson developed the concept of tubes of force in his earlier experimental study of cathode rays and electric discharge (Smith 2001, 27–35; Navarro 2012, 60–70, 114–119). Yet, interference precluded Thomson from assuming that the apparent grainy character of the ether also influenced light.3 Instead, in 1910 and 1913 he advanced two incompatible but equally speculative atomic models that could explain the continuity in the velocity of the emitted electrons. Disregarding Lenard and Ramsauer’s result, both models involved a kind of triggering hypothesis. Suggesting tubes of force inside the atom, Thomson succeeded in regaining a threshold frequency for releasing an electron equivalent to that of Einstein’s equation. Despite the deep differences in their fundamental assumptions, some physicists conflated Thomson’s view of corpuscle-like radiation with Einstein’s, since both ascribed particle-like properties to radiation (Wheaton 1983, 136–142; Stuewer 1970, 252–253; Millikan 1963, 221–223).
Experimental results, however, pointed to a close parallelism between X-rays and light. As mentioned previously, experiments in 1907 showed that like the velocity of electrons released by light, the velocity of electrons released by X-rays is independent of the rays’ intensity, but increases with what was regarded as their hardness. In 1910, Otto Stuhlmann in the United States strengthened the other direction of the analogy. Stuhlmann prepared thin films of platinum by sputtering the material onto quartz plates, which are transparent in ultraviolet light. He showed that similar to the emission induced by X-rays, more electrons were emitted by ultraviolet light in the direction of emergent rather than incident light beam, i.e., more electrons were released in the direction in which light propagated (Stuhlmann 1910; Wheaton 1983, 234–236). In the tradition of experimental physics, these experiments were designed to examine particular claims about the behavior of these rays, rather than to check particular mathematical results. Stuhlmann, for example, tested if ultraviolet beams behaved like X-rays in producing stronger effects in the emergent direction. To that end, he produced special thin platinum surfaces in vacuum and constructed an apparatus that enabled him to compare the effects of emergent and incident beams.
Due to earlier experimental demonstrations of the similarity between different types of radiation, many physicists regarded the celebrated 1912 demonstration of X-ray diffraction merely as an additional support for an already-assumed unity between X-rays and visible light. Following Max Laue’s suggestion, Walter Friedrich and Paul Knipping found interference patterns of X-ray beams passing through crystals. Interference patterns alone, however, could not form conclusive evidence for an undulatory nature. Within a few months, Lorentz showed that like continuous waves impulses also could interfere. Still, this theoretical result did not make much of a difference, especially since the subsequent experiments of William Henry Bragg with his son William Lawrence Bragg, and those of Henry Moseley and Charles G. Darwin on diffraction patterns of X-rays in crystals, showed a full analogy with light waves. “By the fall of 1913, virtually all physicists believed that crystal diffraction had indisputably demonstrated that X-rays are equivalent to ordinary light” (Wheaton 1983, 215). Experiments established the view that the rays are high frequency electromagnetic waves. To demonstrate the wave properties of radiation, William Lawrence Bragg relied on the theoretical analysis of light waves. Still, unlike experiments in the tradition of measurement physics, the conclusion did not depend on the explicit correspondence to a specific mathematical equation (Jenkin 2008, 329–339).
On the one hand, the conclusion apparently solved the quandary about the nature of X- and
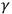 -rays. On the other hand, it highlighted the paradoxical properties of both visible light and X-rays. The rays showed a dual character: a particle-like absorption and a wave-like diffraction. X- and
-rays. On the other hand, it highlighted the paradoxical properties of both visible light and X-rays. The rays showed a dual character: a particle-like absorption and a wave-like diffraction. X- and
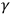 -rays provided a strong qualitative argument against the triggering hypothesis. They led to the implausible assumption that electrons possess an exceedingly high energy inside the atom. The unification of X-rays and light also provided grounds to support Einstein’s relation between the electron’s energy and the frequency of light. In the relatively small range of ultraviolet frequencies, doubts about experimental precision did not allow for a clear conclusion about this relation. Yet, already by 1913, Robert Pohl and Peter Pringsheim had concluded that
-rays provided a strong qualitative argument against the triggering hypothesis. They led to the implausible assumption that electrons possess an exceedingly high energy inside the atom. The unification of X-rays and light also provided grounds to support Einstein’s relation between the electron’s energy and the frequency of light. In the relatively small range of ultraviolet frequencies, doubts about experimental precision did not allow for a clear conclusion about this relation. Yet, already by 1913, Robert Pohl and Peter Pringsheim had concluded that
extrapolation [of the Einstein relation] to the probable frequencies of the Röntgen spectrum leads to velocities for the electron liberated by Röntgen rays which agree in order of magnitude with those experimentally observed. (Pohl and Pringsheim 1913, 1019, emphasis added)
Thus, a rather approximate or qualitative kind of experimental research into the nature of X-rays supported even a theoretical mathematical rule concerning the exact dependence of electron velocity on light frequency.
1.4 Later Theoretical Accounts
By 1911, most physicists had abandoned the triggering hypothesis. Since they had already regarded X-rays as a kind of light before the diffraction experiments, they accepted the evidence from X-rays as supporting Einstein’s linear relation between light frequency and an electron’s energy. Yet they did not accept his explanation. A few of them suggested alternative theories of photoelectricity, deriving the linear relation without admitting “light quanta,” but with a quantum assumption. The failure of the triggering hypothesis and the paradoxes of quantity and quality that discredited an impulse theory, and the theoretical argument from black-body radiation convinced most influential physicists of the need to introduce quantum discontinuity in their theories of the subject. In the background was the success of the quantum hypothesis in accounting for the specific heat of solids at low temperatures (Kuhn 1978, 210–220). Thus, Thomson’s speculative model was the only treatment of the relationship between radiation and matter that did not require directly a quantum assumption, but some sort of structure in the ether through his Faraday tubes and a triggering mechanism.
At the same 1911 meeting in which Ramsauer abandoned the triggering hypothesis, Sommerfeld suggested a promising quantum condition for the ejection of electrons by radiation. According to his suggestion, bound electrons are emitted when the time integral on their Lagrangian (a function of their energy), which increases with the absorption of light, reaches Planck’s quantum of action. In this way, part of their energy originates in the light and part in the atom. With his former assistant Peter Debye, Sommerfeld retrieved Einstein’s equation (without the extraction work) for monochromatic light. Sommerfeld and Debye’s theory, however, suffered from a few problems, the most severe of which turned out to be the accumulation time needed for the release of the first electron. In 1913, barely a month after they published a revised theory, Erich Marx announced his experimental results, according to which the actual time delay was at most 1% of the predicted value (Wheaton 1983, 180–189; Stuewer 1975, 55–58). In the tradition of experimental physics, Marx devised the experiment to examine a specific implication of the theory (shared by a family of electromagnetic theories), rather than to test a mathematical prediction. His experiment practically excluded all explanations that require time accumulation.
In 1913, Planck advanced another explanation of photoelectricity suggesting that a few of the electrons in the atom possess energy close to their emission energy before illumination, so that no accumulation time is needed for their release. Unfortunately, the assumption implied that the electrons’ energy originates in the atoms as in the original triggering hypothesis, and against Lenard and Ramsauer’s finding (Kuhn 1978, 235–254; Wheaton 1983, 178–180).4
The attitude of most physicists toward a theory suggested by Richardson illuminates their positions regarding the interaction between electromagnetic radiation and matter. In 1912, and in a revised version in 1914, Richardson suggested a descriptive rather than an explanatory theory of photoelectricity, “wish[ing] to avoid discussion of the vexed question of the nature of the interaction between the material parts of the system and the æthereal radiation” (quoted in Katzir 2006a, 456). Richardson accounted for the central experimental findings, including Einstein’s equation, by employing thermodynamics and statistical reasoning for equilibrium between emitted and returned electrons, and a quantum assumption through the admission of Planck’s distribution law of electromagnetic radiation. In particular, he did not need to assume the light-quantum hypothesis, which most physicists at the time still rejected. Nevertheless, most physicists did not adopt the new descriptive account. Even if Richardson’s theory suffered from a few internal weaknesses, it seems that for most physicists a descriptive theory, which bypasses the question of mechanism, was not enough to cope with the challenges put forward by the relations between radiation and matter. The failure of classical explanations and the accumulation of evidence from
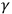 -rays to visible light pointed not only to an unexplained effect, but also to paradoxes. Moreover, unlike many processes of complex matter that were still unexplained, the peculiarities of radiation could not be attributed to an intricate structure whose details were still unknown. Light did not have a complex structure. Advances in the knowledge of atoms suggested that atomic structure was unlikely to account for the interaction of light with matter (Katzir 2006a).
-rays to visible light pointed not only to an unexplained effect, but also to paradoxes. Moreover, unlike many processes of complex matter that were still unexplained, the peculiarities of radiation could not be attributed to an intricate structure whose details were still unknown. Light did not have a complex structure. Advances in the knowledge of atoms suggested that atomic structure was unlikely to account for the interaction of light with matter (Katzir 2006a).
Thus, physicists expected a radical change in the basic concepts of their discipline, and saw little value in attempts to bypass them. Millikan, a strong opponent of the light quantum, spoke for most physicists: “The new facts in the field of radiation […] seem, then, to require in any case a very fundamental revision or extension of classical theories of absorption and emission of radiant energy,” (quoted in Katzir 2006a, 467). These facts included ionization by X- and
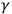 -rays, frequency thresholds and black-body radiation. Most of these facts and their interpretation originated in a study characteristic of a traditional methodology of experimental physics. The challenges posed by the extensive study of different kinds of radiation and their interaction with matter made many scientists more open to ideas and concepts that departed from the classical, regarding not only these but also other physical phenomena.
-rays, frequency thresholds and black-body radiation. Most of these facts and their interpretation originated in a study characteristic of a traditional methodology of experimental physics. The challenges posed by the extensive study of different kinds of radiation and their interaction with matter made many scientists more open to ideas and concepts that departed from the classical, regarding not only these but also other physical phenomena.
1.5 Conclusion
“Experimental physicists,” in the meaning discussed in this paper, discovered photoelectricity, X- and
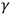 -rays, revealed their surprising properties and exposed the deficiency of established notions and laws of physics to account for them. Neither elaborated theories characteristic of the emerging subdiscipline of theoretical physics nor precise measurements for testing their mathematical predictions drove the research on these phenomena. Rather, physicists working in more traditional modes of research suggested and later rejected the triggering hypothesis, displayed the similitude of visual light, ultraviolet, X- and
-rays, revealed their surprising properties and exposed the deficiency of established notions and laws of physics to account for them. Neither elaborated theories characteristic of the emerging subdiscipline of theoretical physics nor precise measurements for testing their mathematical predictions drove the research on these phenomena. Rather, physicists working in more traditional modes of research suggested and later rejected the triggering hypothesis, displayed the similitude of visual light, ultraviolet, X- and
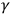 -rays, revealed the paradoxes of quantity and quality and the wave-particle character of these rays. This kind of experimental physics was not detached from theory. On the contrary, theories and assumptions played a crucial role in interpreting experimental results and also in directing research. Theory and experiment were closely connected. Experimentalists explored the empirical implications of particular claims and hypotheses, designing their experiments to that end and suggesting hypotheses toward and following their results.
-rays, revealed the paradoxes of quantity and quality and the wave-particle character of these rays. This kind of experimental physics was not detached from theory. On the contrary, theories and assumptions played a crucial role in interpreting experimental results and also in directing research. Theory and experiment were closely connected. Experimentalists explored the empirical implications of particular claims and hypotheses, designing their experiments to that end and suggesting hypotheses toward and following their results.
Thus, this kind of experimental physics, which is often associated more with the nineteenth than with the twentieth century, also played a significant role in the development of quantum physics. The contribution of this kind of research seems to depend more on the stage of the research than on the date of study. In the examples discussed here, experimental physics shaped fields that were not described by comprehensive theories. In fields described by such a theory, like black-body radiation, even when its foundations were under threat, the new theoretical physics with its associated measuring physics dominated. Theoretical and measurement physics prevailed also in more mundane fields of physics that did not experience a break with classical physics. For example, experimental research of the kind described here led piezoelectric research in the “pre-theoretical phase” of its study, i.e., from its discovery in 1880 until the formulation of a comprehensive theory a decade later. With the formulation of such a mathematical theory, the novel theoretical physics guided the study of the field (Katzir 2006b). Theoretical physics and its associated measuring physics seem to characterize research in “theoretical phases,” i.e., after the formulation of a comprehensive mathematical theory, rather than to be associated especially with a research on microphysics and the quantum. This agrees with Suman Seth’s recent claim in a study of Sommerfeld and his school that theoretical physics extended much beyond the fields that are usually associated with it like microphysics, relativity and quantum theory (Seth 2010, 4). Seth’s claim and my claim here—that quantum mechanics was far from restricted to the new theoretical physics—call for caution in linking the rise of quantum mechanics to that of theoretical physics.
References
Buchwald, Jed (1994). The Creation of Scientific Effects: Heinrich Hertz and Electric Waves. Chicago: The University of Chicago Press.
Büttner, Jochen, Jürgen Renn, J. R. (2003). Exploring the Limits of Classical Physics: Planck, Einstein, and the Structure of a Scientific Revolution. Studies in History and Philosophy of Modern Physics 34: 37-59
Darrigol, Olivier (1992). From c-Numbers to q-Numbers: The Classical Analogy in the History of Quantum Theory. Berkeley: University of California Press.
- (2000). Electrodynamics from Ampère to Einstein. Oxford: Oxford University Press.
Einstein, Albert (1905). Über einen die Erzeugung und Verwandlung des Lichtes betreffenden heuristischen Gesichtspunkt. Annalen der Physik 17: 132-148
Hallwachs, Wilhelm (1916). Lichtelektrizität. In: Handbuch der Radiologie Ed. by Erich Marx. Leipzig: Akademische Verlagsgesellschaft 245-561
Heilbron, John L. (1977). Lectures on the History of Atomic Physics. In: History of Twentieth-Century Physics Ed. by Charles Weiner. New York: Academic Press 40-108
- (1994). The Virtual Oscillator as a Guide to Physics Students Lost in Plato's Cave. Science and Education 3: 177-188
Innes, P. D. (1907). On the Velocity of the Cathode Particles Emitted by Various Metals under the Influence of Röntgen Rays, and Its Bearing on the Theory of Atomic Disintegration. Proceedings of the Royal Society A 79: 442-462
Jenkin, John (2008). William and Lawrence Bragg, Father and Son: The Most Extraordinary Collaboration in Science. Oxford: Oxford University Press.
Joffé, Abram (1907). Eine Bemerkung zu der Arbeit von E. Ladenburg `Über Anfangsgeschwindigkeit und Menge der photoelektrischen Elektronen usw.'. Annalen der Physik 24: 939-940
Jordi Taltavull, Marta (forthcoming) Optical Dispersion at the Crossroads between Classical and Quantum Physics. PhD Diss. Humboldt University
Jungnickel, Christa, Russell McCormmach (1986). Intellectual Mastery of Nature: Theoretical Physics from Ohm to Einstein, Volume 2: The New Mighty Theoretical Physics 1870–1925. Chicago: The University of Chicago Press.
Katzir, Shaul (2006a). Thermodynamic Deduction versus Quantum Revolution: The Failure of Richardson's Theory of the Photoelectric Effect. Annals of Science 63(4): 447-469
- (2006b). The Beginnings of Piezoelectricity: A Study in Mundane Physics. Dordrecht: Springer.
Klein, Martin J. (1963). Einstein's First Paper on Quanta. The Natural Philosopher 2: 57-86
Kuhn, Thomas S. (1978). Black-Body Theory and the Quantum Discontinuity 1894–1912. Oxford: Clarendon Press.
Ladenburg, Erich (1907). Über Anfangsgeschwindigkeit und Menge der photoelektrischen Elektronen in ihrem Zusammenhange mit der Wellenlänge des ausläsenden Lichtes. Physikalische Zeitschrift 8: 590-594
Lienhop, A. (1906). Über die lichtelektrische Wirkung bei tiefer Temperatur. Annalen der Physik 21: 281-304
Millikan, Robert A. (1963). The Electron: Its Isolation and Measurement and the Determination of Some of Its Properties. Chicago: The University of Chicago Press.
Navarro, Jaume (2012). A History of the Electron: J. J. and G. P. Thomson. Cambridge: Cambridge University Press.
Pohl, Robert, Peter Pringsheim (1913). On the Long-Wave Limits of the Normal Photoelectric Effect. Philosophical Magazine 26: 1017-1024
von Schweidler, Egon R. (1898). Über die lichtelektrischen Erscheinungen. Sitzungsberichte der Mathematisch-Naturwissenschaftlichen Classe der Kaiserlichen Akademie der Wissenschaften Wien 107(IIa): 881-909
Sellmeier, Wolfgang (1872). Über die durch die Aetherschwingungen erregten Mitschwingungen der Körpertheilchen und deren Rückwirkung auf die erstern, besonders zur Erklärung der Dispersion und ihrer Anomalien. Annalen der Physik und Chemie 145: 399-421
Seth, Suman (2010). Crafting the Quantum: Arnold Sommerfeld and the Practice of Theory, 1890–1926. Boston: MIT Press.
Smith, George E. (2001). J. J. Thomson and the Electron, 1897–1899. In: Histories of the Electron: the Birth of Microphysics Ed. by Jed Buchwald, Andrew Warwick. Boston: MIT Press 21-76
Steinle, Friedrich (1998). Exploratives vs. theoriebestimmtes Experimentieren: Ampères frühe Arbeiten zum Elektromagnetismus. In: Experimental Essays – Versuche zum Experiment Ed. by Michael Heidelberger, Friedrich Steinle. Baden-Baden: Nomos-Verlag 272-297
Stuewer, Roger H. (1970). Non-Einsteinian Interpretations of the Photoelectric Effect. In: Historical and Philosophical Perspectives of Science Ed. by Roger H. Stuewer. Minneapolis: University of Minnesota Press 246-263
- (1971a). Hertz's Discovery of the Photoelectric Effect. Actes du XIIIe Congrès International d'Histoire des Sciences VI: 35-43
- (1971b). William H. Bragg's Corpuscular Theory of X-Rays and γ-Rays. The British Journal for the History of Science 5: 258-281
- (1975). The Compton Effect: Turning Point in Physics. New York: Science History Publications.
Stuhlmann, Otto (1910). A Difference in the Photoelectric Effect Caused by Incident and Emergent Light. Philosophical Magazine Series 6 20(116): 331-339
Wheaton, Bruce R. (1978). Philipp Lenard and the Photoelectric Effect, 1889–1911. Historical Studies in the Physical Sciences 9: 299-322
- (1983). The Tiger and the Shark: Empirical Roots of Wave-Particle Dualism. Cambridge: Cambridge University Press.
Footnotes
The impulse hypothesis fitted well the corpuscular view of cathode rays: absorption of one corpuscle (electron) leads to the emission of one short electromagnetic impulse—an X-ray. Adoption of the view that cathode rays are a kind of localized concentration of electric charge, or electrons, thus encouraged the acceptance of the impulse hypothesis.
Since energy calculation was based on previous knowledge of electron’s mass and energy, these experiments exemplify the crucial role played by the view that there is only one kind of electron and the determination of its mass and charge.
The separation of X-rays from visible light was based on the assumption that the grains of ether are small enough to be inconsequential for ordinary light.
The theory shared another defect with the triggering hypothesis, namely, it required a complicated atomic structure to account for the continuous velocity spectrum of the emitted electrons, although unlike Lenard, Planck did assume a resonance mechanism.
